Project Vigicard: Tracking drug allergies using the Tezos blockchain
The Tezos Foundation is supporting the Vigicard project, a project to use blockchain technology to help monitor patients subject to drug allergies.
4 minute read
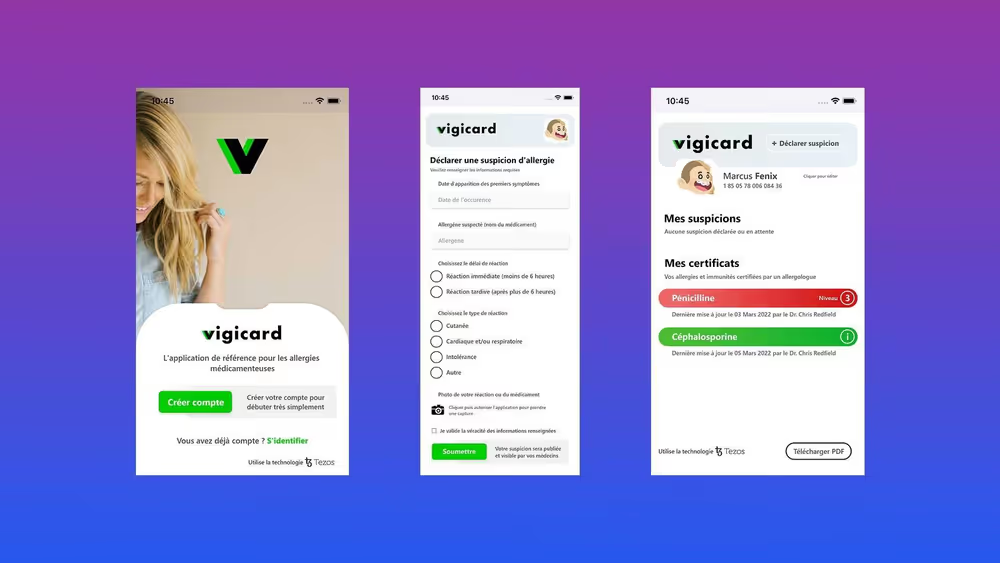
- The Vigicard project is the brainchild of a team of allergists from Montpellier University Hospital and Codinsight
- A clinical trial is planned by the end of the year to better monitor patients following an allergy detection
The Tezos Foundation announces its support for the Vigicard project, the first mobile application improving the monitoring of patients subject to drug allergies. Using Tezos blockchain technology, the application was co-developed by a team of Europe-renowned allergists from Montpellier University Hospital, and Web3 experts Codinsight.
Creating a more serene allergy patient diagnostic pathway #
Developed thanks to research by Dr Anca Chiriac and Prof Pascal Demoly at the Montpellier University Hospital, the Vigicard application responds to a public health issue, namely the lack of information and misuse of potentially allergenic drugs. Declared drug allergies affect 5-10% of the European population. According to recent reports, when allergy tests are carried out correctly, only 20% of declared allergies are confirmed. In the vast majority of cases, it turns out that the drug associated with an allegedly allergic reaction is tolerated by the patient. The Vigicard application will thus enable patients and non-allergist professionals to better identify, treat and manage drug allergies.
Anca Mirela Chiriac, associate professor & allergist behind the project:
“We’re very excited to see the Vigicard project become a reality. Our aim is to use technology to facilitate and considerably improve the monitoring and treatment of patients with drug allergies. In practice, our aim is above all to help practitioners avoid second-line therapeutic prescriptions, which are often less effective and even more toxic, as well as unnecessary avoidance measures.”
To get started, the Vigicard project chose blockchain technology from the outset, notably for its immutability and security of patients’ personal data, while enabling real-time sharing with authorized healthcare professionals. This is particularly crucial in the medical field, where the protection of sensitive information is an absolute priority.
The Tezos blockchain was selected for its intrinsic characteristics of reliability, enhanced security and low-energy self-amending protocol. These qualities are essential to guarantee the stability and longevity of the Vigicard application over time. In addition, the project also benefited from financial support from the Tezos Foundation and technical support from Nomadic Labs, affiliate of the Tezos Foundation and an expert in Tezos blockchain technology, and also from the French company Functori.
Cédric Roche, Adoption Manager France at Nomadic Labs:
“We’re delighted to have supported the project teams in the design and creation of this first digital card. Vigicard is the starting point for a major revolution, both in terms of research and medical monitoring of patients, but also from a technological point of view, with a concrete application of blockchain for the greatest number of people.”
A French project with European ambitions #
Ultimately, the Vigicard project aims to integrate a clinical decision support system (CDSS) for the management of drug allergies. This is a first in the field, supported by the publication of several European directives aimed at improving the quality of care. To validate this integration, the Vigicard application will be tested in a randomized clinical trial in the allergy unit at Montpellier University Hospital. This is the first step in a project that aims to be at the forefront of medicine and patient monitoring.
Fabien Bucamp, co-founder of Codinsight:
“Over and above research, the clinical trial will enable us to determine the usefulness of a decentralized healthcare solution. Indeed, healthcare systems are facing huge problems of interoperability and technological integration, so it’s vital that the project is in tune with these key issues. We’re quite confident, since blockchain has the capacity to meet this need for flexibility to develop large-scale use cases, and to do so securely with existing infrastructures”.
If successful, the application could be rolled out across Europe, and in the future extended to other types of allergy (e.g. food allergies) and health monitoring.